Lewis electron - dot structure
\[\require{mhchem}\]
The Lewis electron-dot model is based on the fact that all atoms combine to give compounds in which each atom or ion achieves the noble gas valence electron configuration of \(s^{2}p^{6}\) (or \(s^{2}\) for hydrogen) in its valence shell, as this is the most stable configuration.
Lewis Structures of Molecular Compounds
Draw the Lewis structure of \(\ce{SO}_{2}\)
Step 1 - Count the total number of valence shell electrons of all the atoms involved in the molecule
Sulphur dioxide has two sulphur atoms and one oxygen atom.
| Atom | Atomic number | Electron configuration | Valence shell electrons |
|---|---|---|---|
| \(\ce{S}\) | \(16\) | \(1s^{2}2s^{2}2p^{6}3s^{2}3p^{4}\) | \(6\) |
| \(\ce{O}\) | \(8\) | \(1s^{2}2s^{2}2p^{4}\) | \(6\) |
Total number of valence shell electrons \(=6+\left(6\times2\right)=18\)
Step 2 - Predict the central atom.
The atom that can form most bonds is considered as the central atom. If two different atoms can form the same number of bonds, then the least electronegative atom should be the central atom. Among sulphur and oxygen, sulphur is the least electronegative atom.
Step 3 - Draw the bonds connected to the central atom.

Step 4 - Add the lone pairs of electrons to the atoms connected to the central atom.
Calculate the remaining valence electrons after drawing bonds. Four valence electrons are used for two bonds.
Remaining valence electrons \(=18-4=14\). These fourteen valence electrons can be distributed among atoms connected to the central atom as lone pairs to complete the octet.
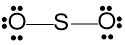
Step 5 - Assign lone pairs of electrons to the central atom if any valence electrons remain.
\(14-12=2\)
To assign lone pairs to the connected atoms, we have used twelve electrons. Therefore, we have two electrons remaining, which can be allocated to the central atom as a lone pair.
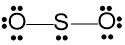
Step 6 - If the central atom still lacks the octet, create multiple bonds with connected atoms using lone pairs of electrons.
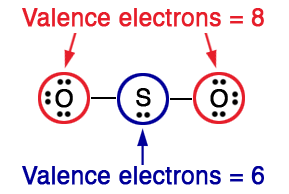
Since sulphur has not acquired the octet, create a double bond with one of the oxygens using a lone pair. With the double bond, all three atoms have acquired an octet. Finally, calculate the formal charges for each atom. Formal charges should be closer to zero for each atom. Use the following formula to calculate the formal charges.
Formal charge \(=\) Valence electrons of the atom \(-\) Lone pairs of electrons \(-\) Bonding electrons

For \(\ce{S}\), formal charge \(=6-2-3=+1\)
For single-bonded \(\ce{O}\), formal charge \(=6-6-1=-1\)
For double-bonded \(\ce{O}\), formal charge \(=6-4-2=0\)
Lewis Structures of Polyatomic Ions
Polyatomic Anions
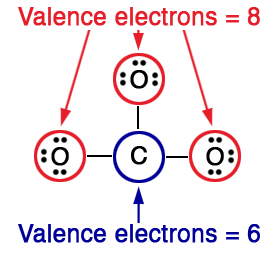
Draw the Lewis structure of \(\ce{CO}_{3}^{2-}\) (carbonate ion)
Step 1 - Count the total number of valence shell electrons of all the atoms involved in the molecule, including the charge of the ion.
Carbonate ion is composed of three oxygen atoms and one carbon atom.
| Atom | Atomic number | Electron configuration | Valence shell electrons |
|---|---|---|---|
| \(\ce{C}\) | \(6\) | \(1s^{2}2s^{2}2p^{2}\) | \(4\) |
| \(\ce{O}\) | \(8\) | \(1s^{2}2s^{2}2p^{4}\) | \(6\) |
Total number of valence electrons\(=4+\left(6\times3\right)+2\)(from the negative charge) \(=24\)
Step 2 - Predict the central atom.
Among carbon and oxygen, carbon can form many bonds.
Step 3 - Draw the bonds connected to the central atom.
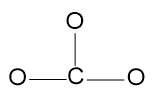
Step 4 - Add the lone pairs of electrons to the atoms connected to the central atom.
Calculate the remaing valence electrons after drawing bonds. Six valence electrons are given for three bonds.
Remaining valence electrons \(=24-6=18\). These eighteen valence electrons can be distributed among atoms connected to the central atom as lone pairs to complete the octet.
.png)
Step 5 - Assign lone pairs of electrons to the central atom if any valence electrons remain.
Not applicable since we have used all the valence electrons.
Step 6 - If the central atom still lacks the octet, create multiple bonds with connected atoms using lone pairs of electrons.
The central atom only has six electrons, therefore create a double bond with one of the oxygen atoms using lone pairs of electrons.
Calculate the formal charges and allocate two negative charges.
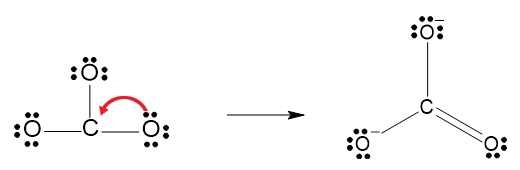
Formal charge \(=\) Valence electrons of the atom \(-\) Lone pairs of electrons \(-\) Bonding electrons
Formal charge for \(\ce{C}\) \(=4-0-4=0\)
Formal charge for double bonded \(\ce{O}\) \(=6-4-2=0\)
Formal charge for single bonded \(\ce{O}\)’s \(=6-6-1=-1\)
Polyatomic Cations
Draw the Lewis structure of \(\ce{NH}_{4}^{+}\) (ammonium ion)
Step 1 - Count the total number of valence shell electrons of all the atoms involved in the molecule, including the charge of the ion.
The ammonium ion is composed of one nitrogen atom and four hydrogen atoms.
| Atom | Atomic number | Electron configuration | Valence shell electrons |
|---|---|---|---|
| \(\ce{N}\) | 7 | \(1s^{2}2s^{2}2p^{3}\) | \(5\) |
| \(\ce{H}\) | \(1\) | \(1s^{1}\) | \(1\) |
Total number of valence electrons\(=5+\left(1\times4\right)-1\)(from the positive charge) \(=8\)
Step 2 - Predict the central atom
Since hydrogen can not handle many bonds, nitrogen is the central atom.
Step 3 - Draw the bonds connected to the central atom.
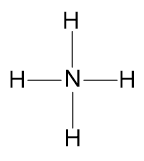
Step 4 - Add the lone pairs of electrons to the atoms connected to the central atom.
Calculate the remaining valence electrons after drawing bonds. Eight valence electrons are given for four bonds.
Remaining valence electrons \(=8-8=0\). Since all the valence electrons have been used no need for step 4.
Step 5 - Assign lone pairs of electrons to the central atom if any valence electrons remain.
Not applicable since all the valence electrons have been used.
Step 6 - If the central atom still lacks the octet, create multiple bonds with connected atoms using lone pairs of electrons.
Since the central atom already acquired the octet, this step is not required.
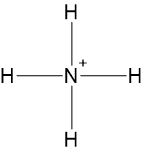
Calculate the formal charges and assign the positive charge.
Formal charge \(=\) Valence electrons of the atom \(-\) Lone pairs of electrons \(-\) Bonding electrons
Formal charge for \(\ce{N}\) \(=5-0-4=+1\)
Formal charge for double-bonded \(\ce{H}\)’s \(=1-0-1=0\)
Exceptions to the Octet Rule
Some molecules do not follow the octet rule. Atoms such as sulphur, phosphorus and chlorine deviate from the octet rule by having more than eight electrons in the valence shell. This is due to the availability of a vacant \(d\) orbital in the third shell.
